As the medical device industry heads into 2026, it finds itself at a pivotal moment shaped by rapid technological change,...
FDA
Food and Drug Administration Commissioner Marty Makary.ANDREW CABALLERO-REYNOLDS/AFP via Getty Images Lizzy Lawrence leads STAT’s coverage of the Food and...
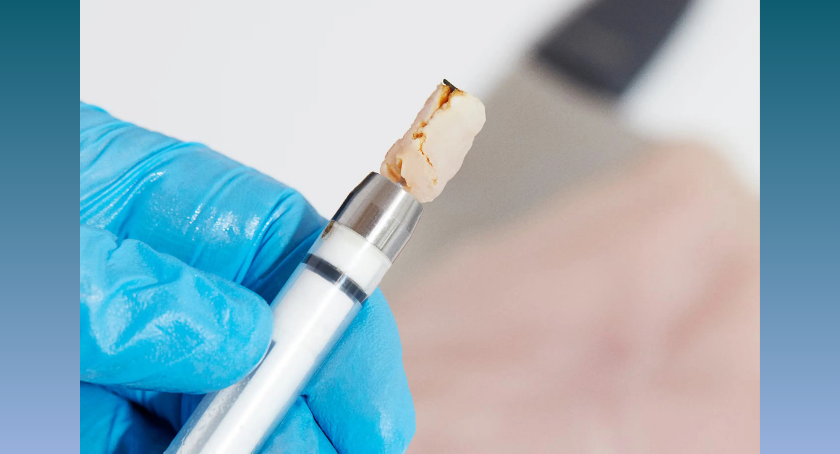
Resitu Medical has received U.S. Food and Drug Administration 510(k) clearance for its RESL09 device, which is used to obtain breast tissue...
Listen to the article 3 min This audio is auto-generated. Please let us know if you have feedback. A warning...